Is Table Sugar a Solute or Solvent
Isopropyl alcohol IPA Acetone. Further if the sugar-water solution is passed through a filter it will be unchanged.
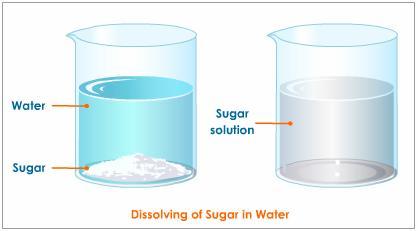
How Would You Identify The Solute And Solvent In This Solution Table Sugar C 12h 22o 11 In Water Socratic
Common examples of solutions are.

. All the components in a solution appear as a single phase. The thing to remember about a solution is that you can distinguish between the solute and the solvent by simply looking at how much of each you have present in the solution. Simply put a solution will always contain more particles of solvent than particles of solute.
The solute may be compounds such as salt sugar carbon dioxide and many more. Steel an alloy of 95 iron 15 carbon and 35 manganese. Sugar solute water solvent sugar water solution.
Now when we prepare sugar water we mix sugar into water and thus sugar is the solute while water is the solvent. When table sugar sucrose dissolves in water is the water the solvent or the solute. Examples include common salt sugar copper II tetraoxosulphate VI etc.
Most solvents are in a liquid state but some solvents might be in a gas or solid-state. In most cases the solvent the one thats dissolving other molecules is water. Here is a list of some common solvents used in chemistry.
Sugar in water carbon dioxide in water and other similar solutions are examples. Sodium chloride NaCl and other salts. This solution is homogeneous because the mixed substances cannot be differentiated.
Examples are water ethanol benzene etc. It can be solid liquid or gas. The solvent is generally.
Here are some common solutes in chemistry. The combination of a solute and a solvent is also called a solution. This is a substance that dissolves in a solvent to form a solution.
Common Solvent Examples. The solvent breaks down the larger solute particle into smaller particles that can then be dispersed throughout the solution. A solvent is a substance that dissolves the solute particles during the formation of a solution.
Identify the solute and solvent in each of the following solutions. The combination of the solute and the solvent produces a mixture that may have different properties than each had before mixing. The SOLUTEs is the substance present in a lesser amount.
There are two types of solutions depending on whether the solvent is water or not. Salt - NaCl - dissolves in water so a solution homogeneous mixture is formed with water as the solvent and salt as the solute. In order to be a true solution a mixture must be stable.
Since a solution is constituted by a liquid solvent where another substance a solute is dissolved table sugar is not a solution. Air a solution of 78 N2 21 O2 and various other gases No solvent O2 solute c a solution of 31 ethanol and 69 water ethano Solute solvent 3 water d. Salt water for example is a direct result of mixing salt with water an example of a single-solute solution whereas a coffee with sugar would be the result of mixing sugar and coffee granules both solutes with a solvent base water.
1 Sugar is solute water is solvent. Neither salt nor sugar dissolve in. This is a substance that dissolves a solute to form a solution.
It can be liquid or gas. Aqueous solutions are those in which the solvent is water. The solute is dissolved from the solvent.
These solvents and solutes may be gases liquids or solids. Cocoa in hot chocolate. The same is true for sugar and water.
Carbon dioxide in sodas. Sugar is the solute and water is the solvent. Table sugar C12H22011 in water table sugar solute water solvent a b.
A solution is defined as a homogeneous mixture of two or more components in which the particle size is smaller than 1 nm. When sugar is fully dissolved into water it can stand for an indefinite amount of time and the sugar will not settle out of the solution.

Solved Identify The Solutes And Solvents For The Following Chegg Com
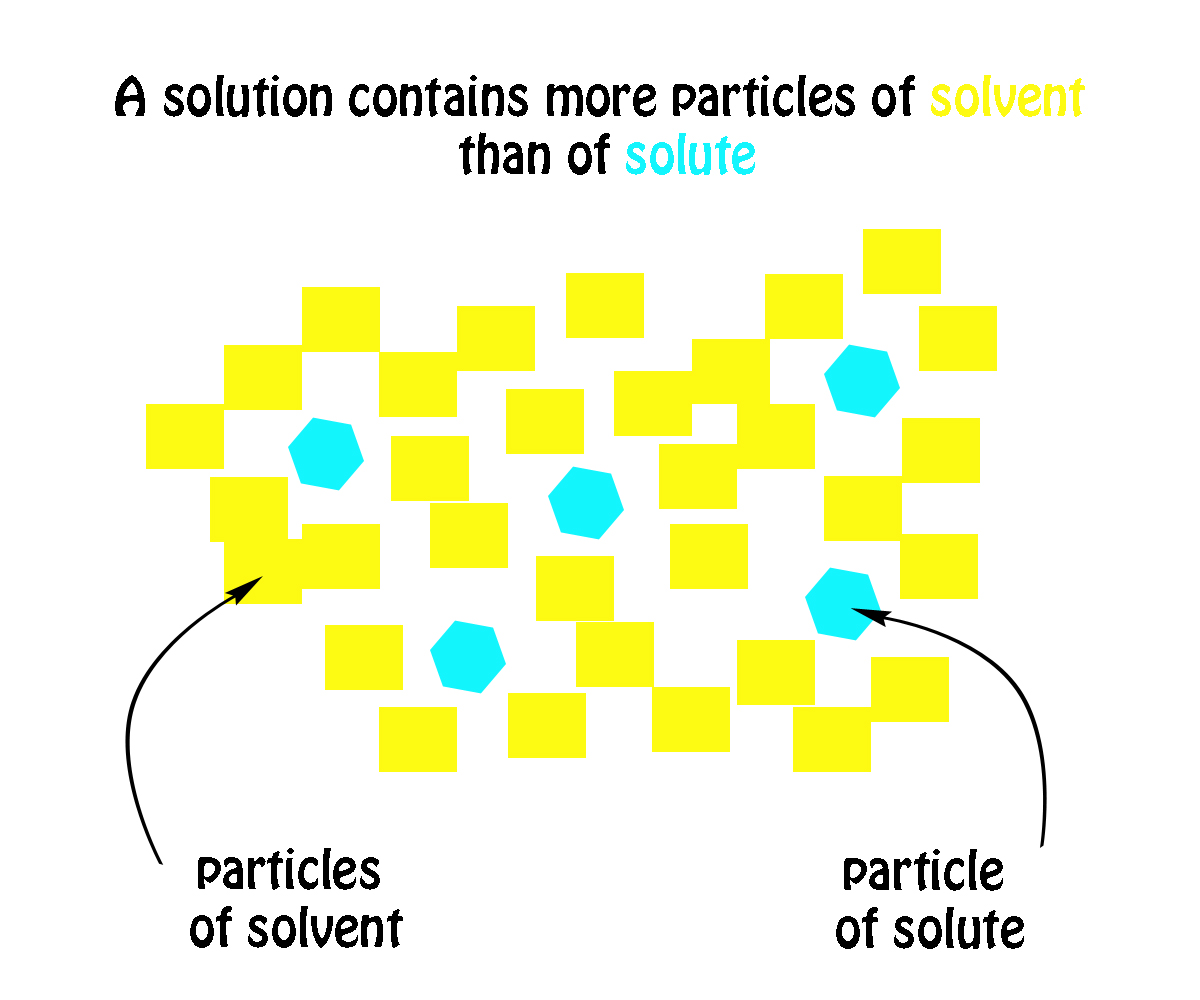
How Would You Identify The Solute And Solvent In This Solution Table Sugar C 12h 22o 11 In Water Socratic

Solved Identify The Solute And Solvent In Each Of The Chegg Com
Comments
Post a Comment